Ultrasound-Assisted Evaluation of the Airway in Clinical Anesthesia Practice: Past, Present and Future
Gausan Ratna Bajracharya, Angela T Truong, Dam-Thuy Truong, Juan Pablo Cata
Gausan Ratna Bajracharya1, Angela T Truong2, Dam-Thuy Truong2 Juan Pablo Cata2,3*
1Department of Anesthesiology and Perioperative Medicine, Clinic-Health Care Centre, Pvt. Ltd., Kathmandu, Nepal
2Department of Anesthesiology and Perioperative Medicine, The University of Texas MD Anderson Cancer Center, Texas, USA
3Anesthesiology and Surgical Oncology Research Group, Texas, USA
- Corresponding Author:
- Juan P. Cata
1515 Holcombe Boulevard, 77030, Houston, USA
Tel: 01 713-7924582
E-mail: jcata@mdanderson.org
Abstract
Introduction: The incidence of difficulties encountered in perioperative airway management has been reported to range from 1% to 4%. In patients with head and neck cancers, the incidence can be dramatically higher. Because of high quality of imaging, non-invasiveness and relatively low cost, ultrasonography has been utilized as a valuable adjunct to the clinical assessment of the airway. A review of the literature was conducted with the objective of summarizing the available evidence concerning the use of ultrasound (US) for assessment of the airway, with special emphasis on head and neck cancers.
Methods and Materials: A systematic search of the literature in the MEDLINE database was performed. A total of 42 manuscripts from 329 searched articles were included in this review.
Results: Ultrasonography was found to give high-resolution images of the anatomic structures of the upper airway comparable to computed tomography and magnetic resonance imaging. Several ultrasonographic parameters (soft tissue thickness at level of hyoid bone, epiglottis and vocal cords, visibility of hyoid bone in sublingual ultrasound, hyomental distance in head-extended position and hyomental distance ratio) were found to be independent predictors of difficult laryngoscopy in obese and non-obese patients. In conjunction with elastosonography, it also provided valuable information regarding tumors, infiltration, and edema as well as fibrosis of the head and neck.
Conclusion: Ultrasound-assisted evaluation of the difficult airway offers many important advantages. The ready availability of US machines in anesthesiology departments, familiarity of anesthesia providers with US guided procedures and the portability of US machines allow real-time, point-of-care assessment. It will undoubtedly become more popular and will greatly contribute to improve perioperative patient safety.
Introduction Implication for Health Policy/Practice/Research/Medical Education
Unanticipated difficult intubations still occur despite the use of clinical predictors. Ultrasound of the upper airway may prove to become a useful adjunct to conventional clinical predictors of difficult airway, especially in patients with head and neck cancers.
Introduction
Securing the airway to ensure alveolar ventilation and prevent pulmonary aspiration constitutes a crucial component in the practice of clinical anesthesia. Yet, adverse respiratory events represent one of the leading causes of malpractice claims for perioperative anesthesia-related incidents [1]. During the induction of general anesthesia, difficulties in achieving adequate ventilation or the inability to perform tracheal intubation may result in the dreaded cannot intubate/cannot ventilate lifethreatening catastrophes [2].
Various risk factors have been identified to help anticipate difficult airways. These include demographic variables (age, gender and race), history of obstructive sleep apnea (OSA), body mass index (BMI), abnormalities of upper teeth, ability to move the lower teeth in front of the upper teeth, inter-incisor gap, modified Mallampati score, thyromental distance, and ability to flex and extend the cervical spine and neck circumference [3]. Despite the widespread adoption of these predictors by anesthesia providers, unexpected difficult ventilation and intubations still continue to occur, reflecting their poor sensitivity, specificity or reproducibility [4]. A pilot study conducted by Adhikari et al. demonstrated that some of these screening tools (Mallampati classification, interincisor gap, thyromental distance and neck mobility) have only poor to moderate sensitivity in predicting a difficult laryngoscopy [5].
Since the introduction of real-time US capabilities, US technology has been adopted and incorporated into daily practice by many medical and surgical specialties. In the operating suites, most anesthesia departments now have at their disposition their own US machines. The ready availability of ultrasound machines in anesthesia departments has promoted its use for vascular access including arterial and central lines, as well as in performance of regional nerve blocks. Using US to help assess the difficult airway constitutes just yet another valuable application of this versatile technology. The newer models are compact and can be rapidly moved with ease from the pre-anesthesia holding area to the operating room. This portability allows point-of-care use of US whenever and wherever it is needed. Since many anesthesia providers had already acquired proficiency in US techniques in US guided vascular access and regional nerve blocks, using US to evaluate the airway could be learned and mastered without too much difficulty. Ultrasound of the upper airway may prove to become a useful adjunct to conventional clinical assessment tools, as it has been successful in visualizing the relevant anatomy and critical structures of the airway [6-8]. The objective of this systematic review is to summarize the literature in regard to the utility of the use of US as an aid to airway assessment, compare and contrast various US parameters that have been studied and suggest potential use of US in the assessment of the airway, in particular, of patients with head and neck cancers.
Material and Methods
Systematic literature search strategies were devised for the MEDLINE database by one of the authors (GRB). Terms used for each concept included both keywords and controlled vocabulary descriptors [ultrasound or ultrasonography or sonography or ultrasonographically or ultrasonographic] and [upper airway or intubation or difficult airway or laryngoscopy or endotracheal tube or endotracheal intubation or tracheal intubation or ventilation] or [ultrasound and imaging and tumors] and [laryngeal or tracheal or epiglottis or tongue or pyriform fossa]. To insure high sensitivity, search strategies were validated for completeness using a pool of known relevant studies. The search included abstracts, case reports, observational studies, controlled clinical trials, guidelines and meta-analysis written in the English language (if any). The search included full texts from December 1979 to April 2015 in human adults age 19 years or older. All searches were scrutinized and selected on the basis of relevance by two independent reviewers (GRB and JPC). Inclusion criteria was set to include articles whose titles or/and abstracts mentioned the use of sonography in upper respiratory tract from the oral cavity to the trachea, its role in general anesthesia and cancers of head and neck, shearwave elastography/elastosonogarphy in head and neck as well as endosonography in assessment of upper respiratory tract malignancies. Articles that described the application of US in lungs and pleura or other organ systems as well as the use of US in the pediatric population were excluded from the study.
Results
A total of 329 articles were retrieved in the first phase of the search. After scrutinizing these articles for relevance, 89 manuscripts were selected for this review and included prospective observational studies, narrative reviews of literature, and letters to the editor, case reports and practice guidelines.
Table 1 summarizes most relevant studies in which US was used to assess the airway. In a feasibility study, Hui et al. suggested that there is an association between an inability to see the hyoid bone on sublingual ultrasound and difficult laryngoscopy view [9]. In that study 11% of the patients were found to have difficult intubation. Clear images of the hyoid bone could be seen in 96.6% of easy intubations while the hyoid bone could not be seen in 72.7% of difficult intubations, indicating a sensitivity and specificity of 73% and 97%, respectively (Table 1). The authors argued that using visibility of the hyoid bone as a surrogate for difficult intubation could not adequately address and evaluate other factors such as tongue size, shape and abnormal tongue [9]. Wojtczak, et al. considered the volume of the tongue and floor of the mouth as well as the hyomental ratio as predictors of difficult laryngoscopy (Table 1) [10]. Hyomental distance ratio was used to denote the change in hyomental distance as the head position changed from neutral position to extended position. In their sample of 12 obese patients whose BMI ranged from 30.1 to 52.3 kg/m2, 50% experienced difficult intubation while the reminding half were easy to intubate. In these two groups, tongue volumes [137 ± 27 cm3 (difficult intubation) versus 168 ± 34 cm3 (easy intubation)] and floor of mouth volumes [34.8 ± 11 cm3 (difficult intubation) versus 37.7 ± 13 cm3 (easy intubation)] did not differ significantly whereas the difference in hyomental distance in head-extended position [52.6 ± 5.8 mm (difficult intubation) versus 65.5 ± 4.1 mm (easy intubation); p<0.01] and hyomental distance ratio [1.02 ± 0.01 (difficult intubation) versus 1.14 ± 0.02 (easy intubation); p<0.002] was significant [10].
| Authors& Year | N= (M/F) Age (mean ± SD) | Difficult/easy laryngoscopy | Parameter studied | Difficult laryngoscopy † | Easy laryngoscopy † | Comment |
|---|---|---|---|---|---|---|
| Ezri, et al. 2003 | 50(21/29) N/A |
9/41 | STT at level of:
|
28 ± 2.7 mm 25 ± 1.3 mm 33 ± 4.3 mm |
17.5 ±1.8 mm 22.8 ± 5 mm 27.4 ± 6.6 mm |
Patients of middle eastern descent with DL have significantly more STT at level of VC. |
| Komatsu, et al. 2007 | 64(12/52) N/A |
20/44 | STT at level of VC* | 20.4 ± 3.0 mm | 22.3 ± 3.8 mm | No clinically significant difference in STT at level of VC could be found between DL and EL groups in patients of Caucasian and African American descent. |
| Adhikari, et al. 2011 | 51(19/32) 53.1 ± 13.2 |
6/45 | STT at level of: • Hyoid bone • Thyrohyoid membrane |
1.69 [95% CI = 1.19-2.19] cm 3.47 [95% CI = 2.88-4.07] cm |
1.37 [95% CI = 1.27-1.46] cm 2.37 [95% CI = 2.29 -2.44] cm |
STT at level of thyrohyoid membrane was significantly more in patients with DL compared to EL |
| Wojtczak, et al. 2012 | 12(7/5) 49.3 ± 15.6 |
6/6 | HMDR** HMDn HMDe Tongue volume Floor of the mouth muscle volume |
1.02 ± 0.01 51.3 ± 5.3 mm 52.6 ± 5.8 mm 137 ± 29 cm3 34.8 ± 11 cm3 |
1.14 ± 0.02 57.5 ± 4.3 mm 65.5 ± 4.1 mm 168 ± 34 cm3 37.7 ± 13 cm3 |
HMDR and HMDe was greater in patients with DL compared to those with EL. |
| Hui, et al. 2014 | 100(55/45) 52.1 ± 15.5 |
11/89 | Hyoid bone not visualized in sublingual ultrasound*** | 8/11(72.7%) | 3/89(3.37%) | Inability to visualize hyoid bone significantly predicted DL. |
| Wu, et al. 2014 | 203(83/120) NA |
28/175 | DSHB*** DSEM*** DSAC*** |
1.51±2.7cm 2.39±0.34cm 1.30±0.31cm |
0.98±0.26cm 1.49±0.39cm 0.92±0.20cm |
DSHB, DSEM and DSAC are independent predictors of DL. |
†Continuous variables are expressed as mean ± standard deviation or mean [95% confidence interval]; categorical variables are expressed as number of occurances (percentage).
Abbreviations: Difficult laryngoscopy (DL), Easy laryngoscopy (EL), Soft tissue thickness (STT), Vocal cords (VC), Hyomental Distance Ratio (HMDR), Hyomental Distance in neck neutral position (HMDn), Hyomental distance in neck extended position (HMDe), Distance from Skin to Hyoid Bone (DSHB), Distance from Skin to Epiglottis Midway between hyoid bone and thyroid cartilage (DSEM), Distance from Skin to Anterior Commissure of vocal cords (DSAC).
Table 1 Ultrasound measurements of anterior neck soft tissue thickness of included studies.
Another study measured the anterior neck soft tissue thickness at five different levels (hyoid bone, thyrohyoid membrane, vocal cords, thyroid isthmus, suprasternal notch) in short axis (transverse view) as well as thickness of the base of the tongue. In that pilot study of 51 subjects, sonographic measurements of anterior neck soft tissue were greater in the difficult laryngoscopy group (Cormack and Lehane grade 3-4), compared to the easy laryngoscopy group (Cormack and Lehane grade 1-2) at the level of the hyoid bone (1.69 [95% CI=1.19 to 2.19] cm versus 1.37 [95% CI=1.27 to 1.46] cm) and thyrohyoid membrane (3.47 [95% CI=2.88 to 4.07] cm versus 2.37 [95% CI=2.29 to 2.44] cm). The subcutaneous thickness of 2.8 cm at the level of the thyrohyoid membrane completely separated the difficult from easy laryngoscopies. No significant correlation was found between sonographic measurement of the thickness of the tongue and Mallampati score as well as interincisor gap [5].
Ezri, et al. measured the distance from the skin to the anterior aspect of the trachea at three levels: zone 1 (vocal cords), zone 2 (thyroid isthmus), and zone 3 (suprasternal notch) by a mean of soft tissue thickness obtained in central axis and 1.5 mm to the left and right of the central axis in 50 morbidly obese patients. There were 18% of cases with difficult laryngoscopy (Cormack and Lehane grade 3 or 4) (Table 1). These patients had much more soft tissue in zone 1 [28 ± 2.7 mm versus 17 ± 1.8 mm, p>0.001] and zone 3 [33 ± 4.3 mm versus 27.4 ± 6.6, p<0.013]. It was concluded that among these, the zone 1 soft tissue thickness appeared to be the better predictor of a difficult laryngoscopy as there was no overlap in its range in the difficult laryngoscopy group (24-32 mm) and the easy laryngoscopy group (15-22 mm) [11]. In a study that included 64 obese patients (BMI>35 kg/ m2) undergoing tracheal intubation under general anesthesia, it was found that patients with difficult laryngoscopy had thinner neck soft tissue at the level of the vocal cords [20.4 ± 3.0 mm (difficult laryngoscopy) versus 22.3 ± 3.8 mm (easy laryngoscopy); p=0.049] [12].
A prospective observational study by Wu, et al. among 203 nonobese patients of Chinese Han population undergoing elective surgeries under general anesthesia evaluated 3 ultrasound parameters: soft tissue thickness at the level of hyoid bone [minimal distance from the hyoid bone to skin surface (DSHB)], at level of thyrohyoid membrane [distance from skin to epiglottis midway (DSEM) between hyoid bone and thyroid cartilage] and the minimal distance from skin to anterior commissure (DSAC) of vocal cords [13]. It was noted that 13.8% of patients had difficult laryngoscopy (Cormack and Lehane grade 3 or 4) and they were found to have greater thickness of anterior neck soft tissue measured by US at the level of the hyoid bone [DSHB (1.51 ± 2.7 cm versus 0.98 ± 0.26 cm; p<0.0001)], thyrohyoid membrane [DSEM (2.39 ± 0.34 cm versus 1.49 ± 0.39 cm; p<0.00010] and anterior commissure [DSAC (1.30 ± 0.31cm versus 0.92 ± 0.20 cm; p<0.0001)] compared to the easy laryngoscopy groups. Using the Youden index, they also calculated the optimal cutoff values with their sensitivity and specificity in parenthesis as: DSHB [1.28 cm (85.7%, 85.1%)], DSEM [1.78 cm (100%, 66.3%] and DSAC [1.1 cm (75.0%, 80.6%)].
US imaging can contribute to anatomic information that would otherwise not be evident on routine clinical screening tests for assessment of a difficult laryngoscopy for instance, thickness of anterior soft tissue, fibrosis or contractures, edema and tumors [5,14-21]. Although computerized tomography (CT) and magnetic resonance imaging (MRI) can provide accurate clinical information, they are costly, time consuming and may impose certain risks to patients including exposure to ionizing radiation and contrast allergies [11]. A study by Prasad, et al. compared images and measurements acquired by US and CT and concluded that all structures visualized by CT can also be reliably identified by using US imaging [22]. Similarly, the measurements provided by US are also comparable to those provided by MRI [23].
Summarizing, in non-obese and obese patients, visibility of the hyoid bone in sublingual US, soft tissue thickness at the level of the hyoid bone, epiglottis, vocal cords and suprasternal notch as well as distance between skin to mid epiglottis at the level of the thyrohyoid membrane are independent predictors of difficult laryngoscopy (Cormack-Lehane grade 3 and 4). However, additional studies in larger patient populations with stratified analysis of age, gender and race need to be carried out in order to validate the use of US as a predictive tool for difficult airway in these patient groups.
Discussion
Ultrasonography and its approaches to the airway
The introduction of anatomical evaluation with the help of US imaging has been one of the most exciting recent technological advances in the field of anesthesia. The increasing use of US has been attributed to its proven clinical efficacy, cost effectiveness and practicality as it allows anesthesiologists to evaluate complex and varied anatomy [24]. US imaging is safe (nonionizing radiation, no contrast agent), non-invasive, portable, widely available, painless, easily reproducible, and gives realtime dynamic images [8,25-27]. Although considered much safer compared to other imaging modalities, exposure in terms of intensity and time should be limited as much as possible, as highenergy US can cause heating and damage to tissues [24].
The scientific principle of ultrasound imaging is based on the generation of high frequency sound waves beyond the audible range (20 KHz) varying from 2-15 MHz that penetrate different tissues of the body at various velocities. Reflected waves from the tissue interface are received by the transducer and analyzed by a computer to produce images. The differences in velocity of US as it passes through normal tissue and air columns generate a characteristic bright hyperechoic linear appearance at airmucosa (A-M) interface in the respiratory tract along with comet tail and reverberation artifacts [6]. Structures that lie behind the column of air are completely obscured [28,29]. Bony structures also appear as a bright hyperechoic line with an acoustic shadow behind them, while muscles and connective tissue membranes appear hypoechoic with a heterogeneous striated appearance. Depending upon the fat content of their parenchyma, glandular structures such as the thyroid gland and submandibular gland appear mildly to strongly hyperechoic with a homogenous appearance.
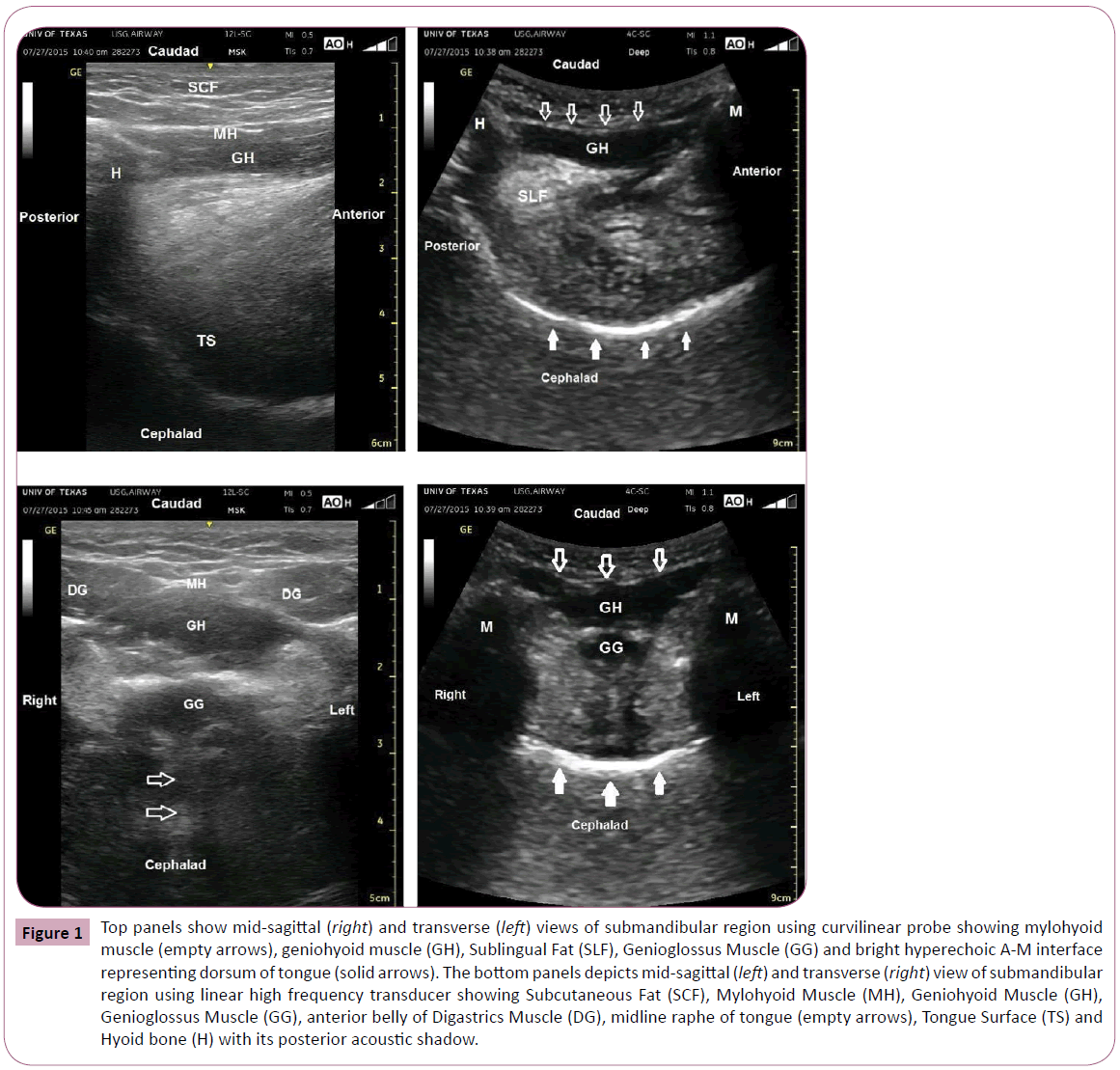
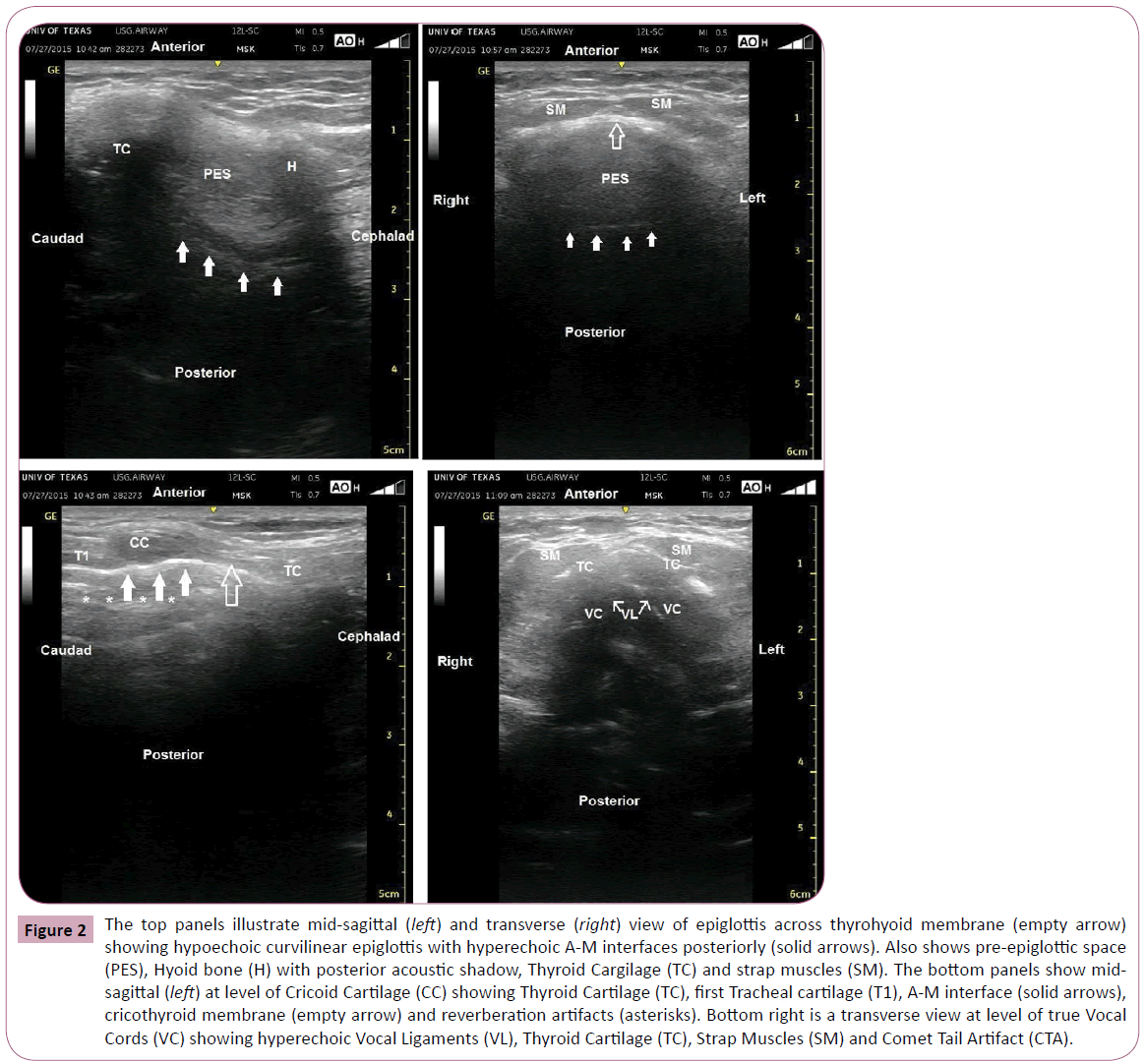
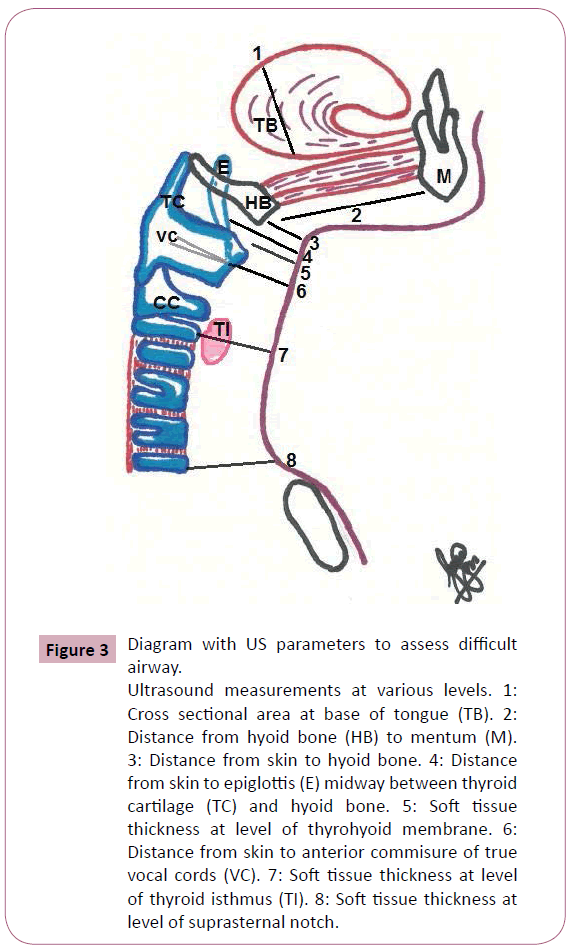
The transcutaneous and the sublingual approach have been described for visualizing the upper airway sonographically. Sublingual US is a well-tolerated, simple and efficient method for preoperative airway assessment in which a single smallfootprint, high-frequency (4-10 MHz) curved array probe with a sterile ultrasound probe cover without conductive gel is used [9]. The transcutaneous scanning approach is preferable as the epiglottis and laryngeal inlet is often not visualized by the acoustic shadowing cast by the hyoid bone and the intraluminal air in the sublingual approach [6]. For transcutaneous scanning, it is recommended that the airway be systematically imaged starting from the mentum coursing along the floor of the mouth to the level of suprasternal notch [6]. Thus, for the purpose of sonographic assessment, the upper airway has been divided into supra-hyoid and infra-hyoid regions (Figures 1 and 2). Multiple studies have concluded that a curved low-frequency (2-5 MHz) transducer can be used to visualize the deeper structures in the sub-mandibular and supraglottic regions due to its wider field of view, while the linear high-frequency (5-14 MHz) transducer can be used to visualize more superficial structures located 2-5 cm from the skin surface [5,6,10,11]. Depending upon the structure of interest, midsagittal, coronal, parasagittal or transverse views are employed [5,8,10]. As demonstrated in Figure 1, several structures can be identified in the suprahyioid region including the mylohyoid, geniohyoid and genioglossus muscles the tongue and hyoid bone. The epiglottis, thyrohyoid membrane, pre-epiglottic space (PES), hyoid bone thyroid cartilage (TC), strap muscles (SM), cricoid cartilage (CC), thyroid cartilage (TC) and first tracheal cartilages are structures of the infrahyoid region (Figure 2). Using these anatomical landmarks, various ultrasonographic parameters (Table 2) have been investigated as indicators of difficult laryngoscopy in obese and non-obese patients; however the most appropriate sonographic indicator predicting difficult laryngoscopy and intubation in patients with cancers of head and neck is yet to be explored [5,6,9-11,13,30]. The soft tissue thickness at 5 different levels (hyoid bone, thyrohyoid membrane, and thyroid isthmus and suprasternal notch) has been described as parameters to predict difficult intubations in obese patients (Figure 3). Other parameters also used by different authors include the tongue volume, hyomental distance and floor of mouth volume (Table 2 and Figure 3).
| Ultrasound parameters | Measurement | Ultrasound landmarks |
|---|---|---|
| Soft tissue thickness at level of hyoid bone | measured from skin to anterior surface of hyoid bone in short axis | Hyoid bone appears as a hyperechoic line with posterior acoustic shadow. |
| Soft tissue thickness at level of thyrohyoid membrane | Measured from skin to anterior surface of epiglottis midway between hyoid bone and thyroid cartilage | Epiglottis appears as a hypoechoic curvilinear line in short axis with hyperechoic A-M interface at its posterior surface and hyperechoic pre-epiglottis space anteriorly |
| Soft tissue thickness at level of true vocal cords | Measured from skin to anterior commisure of true vocal cords | True vocal chords appear as triangular hypoechoic structures with hyperechoic vocal ligaments at their medial border. Anterior commisure is the point where the true vocal cords meet anteriorly. |
| Soft tissue thickness at level of thyroid isthmus | Measured from skin to anterior surface of tracheal cartilage at level of thyroid isthmus | Thyroid isthmus appears as homogenous hyperchoic band of tissue joining thyroid glands. Tracheal cartilage appears as hypoechoic inverted U shaped structure with a linear hyperechoic A-M interface and reverberation artifact posteriorly |
| Soft tissue thickness at level of suprasternal notch | Measured from skin to anterior surface of tracheal cartilage at the suprasternal notch | Tracheal cartilage is identified as hypoechoic inverted U structure with hyperechoic line posteriorly. |
| Tongue volume | Calculated by multiplying the mid-sagittal cross-section area of the tongue by its width (distance between the most distant points on the dorsal surface in mid transverse scan) | The tongue extends from upper surface of hypoechoicgeniohyoid muscle to hyperechoic A-M interface on the dorsal surface of tongue. Intrinsic muscles of tongue give a striated appearance. |
| Hyomental distance | Measured from upper border of hyoid bone to lower border of mentum in head extended and neutral positions. | Mentum appears as hyperechoic line with posterior acoustic shadow in anterior aspect of submandibular region. |
| Floor of mouth volume | Calculated by multiplying the mid-sagittal cross-section area of the floor of mouth by the hyomental distance in neutral position of the head. | Muscles of floor of mouth appear as thick hypoechoic band of geniohyoid muscle lying above the thin hypoechoic band of mylohyoid muscle in midsagittal plane. |
Table 2 Measurement of ultrasound parameters and description of landmarks.
Figure 1: Top panels show mid-sagittal (right) and transverse (left) views of submandibular region using curvilinear probe showing mylohyoid muscle (empty arrows), geniohyoid muscle (GH), Sublingual Fat (SLF), Genioglossus Muscle (GG) and bright hyperechoic A-M interface representing dorsum of tongue (solid arrows). The bottom panels depicts mid-sagittal (left) and transverse (right) view of submandibular region using linear high frequency transducer showing Subcutaneous Fat (SCF), Mylohyoid Muscle (MH), Geniohyoid Muscle (GH), Genioglossus Muscle (GG), anterior belly of Digastrics Muscle (DG), midline raphe of tongue (empty arrows), Tongue Surface (TS) and Hyoid bone (H) with its posterior acoustic shadow.
Figure 2: The top panels illustrate mid-sagittal (left) and transverse (right) view of epiglottis across thyrohyoid membrane (empty arrow) showing hypoechoic curvilinear epiglottis with hyperechoic A-M interfaces posteriorly (solid arrows). Also shows pre-epiglottic space (PES), Hyoid bone (H) with posterior acoustic shadow, Thyroid Cargilage (TC) and strap muscles (SM). The bottom panels show midsagittal (left) at level of Cricoid Cartilage (CC) showing Thyroid Cartilage (TC), first Tracheal cartilage (T1), A-M interface (solid arrows), cricothyroid membrane (empty arrow) and reverberation artifacts (asterisks). Bottom right is a transverse view at level of true Vocal Cords (VC) showing hyperechoic Vocal Ligaments (VL), Thyroid Cartilage (TC), Strap Muscles (SM) and Comet Tail Artifact (CTA).
Figure 3: Diagram with US parameters to assess difficult airway.
Ultrasound measurements at various levels. 1: Cross sectional area at base of tongue (TB). 2: Distance from hyoid bone (HB) to mentum (M). 3: Distance from skin to hyoid bone. 4: Distance from skin to epiglottis (E) midway between thyroid cartilage (TC) and hyoid bone. 5: Soft tissue thickness at level of thyrohyoid membrane. 6: Distance from skin to anterior commisure of true vocal cords (VC). 7: Soft tissue thickness at level of thyroid isthmus (TI). 8: Soft tissue thickness at level of suprasternal notch.
Ultrasonography and cancers of head and neck
Ultrasonography has been used as a diagnostic tool for cancers of the head and neck [14]. Tumors arising in the tongue appear as heterogenous hypoechoic masses infiltrating and distorting the normal fan shaped architecture as well as median raphe of the tongue [31]. Real time ultrasonography can also help in identifying tumor infiltration into adjacent structures by demonstrating restricted mobility of the tongue [32]. Any increase in tongue volume as well as limited mobility and stiffness secondary to the tumor itself or its infiltration can contribute to difficulty in displacement of the tongue during direct laryngoscopy and visualization of the glottis.
Using vocal cords as landmarks, laryngeal tumors appearing as a nodule or mass can be identified as supraglottic, gottic or subglottic depending upon the site of the maximal portion [14,33]. Any invasion of extra laryngeal structures including epiglottis, ventricular bands, vocal cords, anterior commisure, subglottis, pre-epiglottic and paraglottic spaces, thyroid cartilage, thyroid gland and cervical soft tissues appears as a nodule, mass or a thickened change [34,35]. US was found to have a sensitivity of 80% in evaluating invasion of intra-and extra laryngeal structures in the glottis and supraglottic region and 72.7% in subglottic region, and a specificity of 90% in all regions [35]. Invasion of the paraglottic space is seen as hypoechoic tumor intruding into or interrupting the linear hyperechoic fatty layer as well as decreased or absent relative movement of the inner perichondrium of thyroid cartilage [15,34]. Erosion of the thyroid cartilage can be seen as an interruption of the inner and/or outer hyperechoic lines that represent perichondrium and replacement of the central medulla by hypoechoic tumors [14,34,35]. Asymmetrical movement or immobile status of the vocal cords or arytenoid cartilages during speaking or breathing could denote laryngeal fixation due to tumor infiltration; in addition to these features, the shape, margin, echogenicity and vascularity of tumors can be evaluated with ultrasonography [14,33,15].
Any mass protruding into the airway lumen may be clearly visualized as it partially or completely displaces the intraluminal air removing the artifact it should have created [33,15,36]. In addition, US can provide accurate measurement of the available glottic space and the diameter of the trachea in order to select the most appropriate size of endotracheal tube for intubation [30,36-38]. It can also be used in measurement of soft tissue thickness at various levels of the upper respiratory tract especially in the areas that need to be displaced by the laryngoscope blade during direct laryngoscopy as an indicator for difficult intubation [5,13].
In cases in which the larynx and trachea cannot be assessed due to thickening and fibrosis of the subcutaneous tissues of the neck, US can aid in identifying and localizing these structures when simple palpation cannot [8]. US technology using elastosonography can also be used to identify qualitative and quantitative fibrotic changes in muscles, connective tissue and skin. This is of particular value in patients with fibrosis as a side effect of radiotherapy [39]. This method of determination of propagation velocity of the shear waves (SWV) using Acoustic Radiation Force Impulse method (ARFI) has also been used in evaluating oncological pathologies involving lymph nodes, thyroid and salivary glands and other soft tissue tumors [40-43,44,45]. In this light, elastosonography can be an important area for future research in quantifying stiffness of muscles and soft tissues of the floor of the mouth which can aid in predicting difficult intubation in patients with irradiated necks.
The difficult airway scenarios most likely to require US-assisted evaluation are impossible to assess using clinical predictors alone. Difficult airway cases include patients who have undergone previous major composite resections for head/neck cancers followed by plastic reconstruction. The presence of these complex plastic reconstructions with bone and/or soft tissue free flaps makes airway assessment by conventional physical examination alone very difficult [45]. Post-surgical changes are often compounded by radiation changes which results in severe anatomic distortion [3]. In these cases, it may be impossible to recognize anatomic landmarks of the airway during routine laryngoscopy. US can allow for more precise assessment of airway structures and can help the anesthesiologist to formulate a safe plan for securing the airway, such as choosing to perform an awake fiberoptic intubation in the case of complete destruction of the epiglottis by prior surgical resection and radiation.
Other pathologies that US can help to delineate the location and degree of airway invasiveness include advanced oropharyngeal cancers with extension to vallecular and supraglottic area, epiglottic tumors, and carcinomas of the vocal cords [14,15,33]. US can help visualize these lower airway structures and provide valuable information to the anesthesiologist prior to airway manipulation. It is worth mentioning that volume of the thyroid gland failed as predictor of difficult intubation in patients who had thyroid surgery [46]. Patients who present with massive deep neck infections such as retropharyngeal abscess or Ludwig’s angina may also potentially benefit from US airway assessment. These are often very challenging cases for airway control. US can be used to assess the abscess and the risks of airway obstruction and abscess rupture during airway manipulation [47]
If the abscess is seen to have a very thick wall, well-sealed off and with low risk of rupture, then one may choose to perform an awake fiberoptic intubation. However, if a very thin-walled abscess with friable tissues is seen by US, then the most prudent option would be to proceed with an awake tracheostomy by the head and neck surgeon.
In patients with severe trismus not allowing passage of tracheal tubes between the teeth and concomittent contraindications to nasal intubation, flexible fiberoptic retromolar intubation may be selected to secure the airway [48]. In these cases, US imaging could permit the precise evaluation of the dimensions of the retromolar space bilaterally. The size of the tracheal tube and the side with the larger space may be chosen accordingly for intubation.
Conclusion
Ultrasound-assisted evaluation of the airway performed by the attending anesthesiologist has important advantages compared to clinical assessment and to other modalities such as CT scan and MRI. In contrast to CT and MRI scans which may have been done many days previously and during this interval, the tumor may have grown significantly larger, US provides real-time information necessary to make help select the best approach to secure the airway. Hence, US of the upper airway may become a useful tool to evaluate difficult airways, especially in patients with head and neck cancers. Unfortunately, this last statement will remain merely speculative until well-conducted studies are performed to test the hypothesis that the use US in patients with head and neck cancer facilitates clinical decisions on the management of difficult airways.
References
- Met zner J, Posner KL, Lam MS, Domino KB (2011) Closed claims' analysis. Best Pract Res ClinAnaesthesiol 25: 263-276.
- Schaeuble JC, Ganter MT (2015) Clarifying the indications for difficult airway alert forms. Anaesthesia 70: 505-506.
- Kheterpal S, Healy D, Aziz MF, Shanks AM, Freundlich RE, et al. (2013) Incidence, predictors, and outcome of difficult mask ventilation combined with difficult laryngoscopy: a report from the multicenter perioperative outcomes group. Anesthesiology 119: 1360-1369.
- Vannucci A, Cavallone LF (2015) Bedside predictors of difficult intubation: a systematic review. Minerva Anestesiol.
- Adhikari S, Zeger W, Schmier C, Crum T, Craven A, et al. (2011) Pilot study to determine the utility of point-of-care ultrasound in the assessment of difficult laryngoscopy. AcadEmerg Med 18: 754-758.
- Singh M, Chin KJ, Chan VW, Wong DT, Prasad GA, et al. (2010) Use of sonography for airway assessment: an observational study. J Ultrasound Med 29: 79-85.
- Fiadjoe JE, Stricker P, Gurnaney H, Nishisaki A, Rabinowitz A, et al. (2012) Ultrasound-guided tracheal intubation: a novel intubation technique. Anesthesiology 117: 1389-1391.
- Kristensen MS, Teoh WH, Graumann O, Laursen CB (2014) Ultrasonography for clinical decision-making and intervention in airway management: from the mouth to the lungs and pleurae. Insights Imaging 5: 253-279.
- Hui CM, Tsui BC (2014) Sublingual ultrasound as an assessment method for predicting difficult intubation: a pilot study. Anaesthesia 69: 314-319.
- Wojtczak JA. Submandibular sonography (2012) assessment of hyomental distances and ratio, tongue size, and floor of the mouth musculature using portable sonography. J Ultrasound Med 31: 523-8.
- Ezri T, Gewürtz G, Sessler DI, Medalion B, Szmuk P, et al. (2003) Prediction of difficult laryngoscopy in obese patients by ultrasound quantification of anterior neck soft tissue. Anaesthesia 58: 1111-1114.
- Komatsu R, Sengupta P, Wadhwa A, Akça O, Sessler DI, et al. (2007) Ultrasound quantification of anterior soft tissue thickness fails to predict difficult laryngoscopy in obese patients. Anaesth Intensive Care 35: 32-37.
- Wu J, Dong J, Ding Y, Zheng J (2014) Role of anterior neck soft tissue quantifications by ultrasound in predicting difficult laryngoscopy. Med SciMonit 20: 2343-2350.
- Arens C, Glanz H (1999) Endoscopic high-frequency ultrasound of the larynx. Eur Arch Otorhinolaryngol 256: 316-322.
- Xia CX, Zhu Q, Cheng Y, Zhao HX, Jin ZZ (2011) Sonographic assessment of hypopharyngeal carcinoma: preliminary study. J Ultrasound Med 30: 217-225.
- Narendra PL, Vishal NS, Jenkins B (2014) Ludwig's angina: need for including airways and larynx in ultrasound evaluation. BMJ Case Rep 2014.
- Sutherasan Y, Theerawit P, Hongphanut T, Kiatboonsri C, Kiatboonsri S (2013) Predicting laryngeal edema in intubated patients by portable intensive care unit ultrasound. J Crit Care 28: 675-680.
- Bressmann T, Ackloo E, Heng CL, Irish JC (2007) Quantitative three-dimensional ultrasound imaging of partially resected tongues. Otolaryngol Head Neck Surg 136: 799-805.
- Dhoot NM, Singh S, Choudhury B, Kataki AC, Kalita NK, et al. (2014) Evaluation of hypopharyngeal carcinoma using high-resolution ultrasound: comparison with CT. J Clin Ultrasound 42: 143-149.
- Fakhry C, Agrawal N, Califano J, Messing B, Liu J, et al. (2014) The use of ultrasound in the search for the primary site of unknown primary head and neck squamous cell cancers. Oral Oncol 50: 640-645.
- Zhou J, Zhu SY, Liu RC, Luo F, Shu DX (2009) Vascularity index of laryngeal cancer derived from 3-D ultrasound: a predicting factor for the in vivo assessment of cervical lymph node status. Ultrasound Med Biol 35: 1596-1600.
- Prasad A, Yu E, Wong DT, Karkhanis R, Gullane P, et al. (2011) Comparison of sonography and computed tomography as imaging tools for assessment of airway structures. J Ultrasound Med 30: 965-972.
- Lakhal K, Delplace X, Cottier JP, Tranquart F, Sauvagnac X, et al. (2007) The feasibility of ultrasound to assess subglottic diameter. AnesthAnalg 104: 611-614.
- Gupta PK, Gupta K, Dwivedi AN, Jain M (2011) Potential role of ultrasound in anesthesia and intensive care. Anesth Essays Res 5: 11-19.
- Adi O, Chuan TW, Rishya M (2013) A feasibility study on bedside upper airway ultrasonography compared to waveform capnography for verifying endotracheal tube location after intubation. Crit Ultrasound J 5: 7.
- Al-Abed MA, Antich P, Watenpaugh DE, Behbehani K (2012) Upper airway occlusion detection using a novel ultrasound technique. ConfProc IEEE Eng Med BiolSoc 2012: 5650-5653.
- Ding LW, Wang HC, Wu HD, Chang CJ, Yang PC (2006) Laryngeal ultrasound: a useful method in predicting post-extubation stridor. A pilot study. EurRespir J 27: 384-389.
- Tsui BC, Hui CM (2009) Challenges in sublingual airway ultrasound interpretation. Can J Anaesth 56: 393-394.
- Abernethy LJ, Allan PL, Drummond GB (1990) Ultrasound assessment of the position of the tongue during induction of anaesthesia. Br J Anaesth 65: 744-748.
- TurkayAydogmus M, Erkalp K, Nadir Sinikoglu S, Usta TA, G OU, et al (2014) Is ultrasonic investigation of transverse tracheal air shadow diameter reasonable for evaluation of difficult airway in pregnant women: A prospective comparative study. Pak J Med Sci 30: 91-95.
- Mettler FA Jr, Schultz K, Kelsey CA, Khan K, Sala J, et al. (1979) Gray-scale ultrasonography in the evaluation of neoplastic invasion of the base of the tongue. Radiology 133: 781-784.
- Yesuratnam A, Wiesenfeld D, Tsui A, Iseli TA, Hoorn SV, et al. (2014) Preoperative evaluation of oral tongue squamous cell carcinoma with intraoral ultrasound and magnetic resonance imaging-comparison with histopathological tumour thickness and accuracy in guiding patient management. Int J Oral MaxillofacSurg 43: 787-794.
- Xia CX, Zhu Q, Zhao HX, Yan F, Li SL, et al. (2013) Usefulness of ultrasonography in assessment of laryngeal carcinoma. Br J Radiol 86: 20130343.
- Kraft M, Bruns N, Hügens-Penzel M, Arens C (2013) Clinical value of endosonography in the assessment of laryngeal cancer. Head Neck 35: 195-200.
- Rothberg R, Noyek AM, Freeman JL, Steinhardt MI, Stoll S, et al. (1986) Thyroid cartilage imaging with diagnostic ultrasound. Correlative studies. Arch Otolaryngol Head Neck Surg 112: 503-515.
- Raghavendra BN, Horii SC, Reede DL, Rumancik WM, Persky M, et al. (1987) Sonographic anatomy of the larynx, with particular reference to the vocal cords. J Ultrasound Med 6: 225-230.
- Bright L, Secko M, Mehta N, Paladino L, Sinert R (2014) Is there a correlation of sonographic measurements of true vocal cords with gender or body mass indices in normal healthy volunteers? J Emerg Trauma Shock. 7: 112-115.
- Badea I, Tamas-Szora A, Chiorean I, Fildan F, Ciulea E, et al. (2014) Quantitative assessment of the masseter muscle's elasticity using Acoustic Radiation Force Impulse. Med Ultrason 16: 89-94.
- Choi YJ, Lee JH, Lim HK, Kim SY, Han MW, et al. (2013) Quantitative shear wave elastography in the evaluation of metastatic cervical lymph nodes. Ultrasound Med Biol 39: 935-940.
- Cakir B, Aydin C, KorukluoÄŸlu B, Ozdemir D, Sisman IC, et al. (2011) Diagnostic value of elastosonographically determined strain index in the differential diagnosis of benign and malignant thyroid nodules. Endocrine 39: 89-98.
- Wierzbicka M, Kauny J, Szczepanek-Parulska E, Stangierski A, Gurgul E, et al. (2013) Is sonoelastography a helpful method for evaluation of parotid tumors? Eur Arch Otorhinolaryngol 270: 2101-2107.
- Kauny J, Kope T, Szczepanek-Parulska E, Stangierski A, Gurgul E, et al. (2014) Shear wave elastography: a new noninvasive tool to assess the intensity of fibrosis of irradiated salivary glands in head and neck cancer patients. Biomed Res Int 2014: 157809.
- Hata H, Natsuga K, Aoyagi S, Homma E, Shimizu H (2014) Ultrasound B-mode and elastographic findings of angiomatoid fibrous histiocytoma. ClinExpDermatol 39: 538-539.
- Soydan SS, Bayram B, Akdeniz BS, Kayhan Z, Uckan S (2015) Changes in difficult airway predictors following mandibular setback surgery. Int J Oral Maxillofac Surg.
- Meco BC, Alanoglu Z, Yilmaz AA, Basaran C, Alkis N, et al. (2015) Does ultrasonographic volume of the thyroid gland correlate with difficult intubation? An observational study. Brazilian journal of anesthesiology 65: 230-234.
- el-Sayed Y, al Dousary S (1996) Deep-neck space abscesses. J Otolaryngol 25: 227-233.
- Truong AT, Ahmad B, Cata JP, Martin JW, Truong DT, et al. (2015) Measurement of retromolar space dimensions using dental pantomograms for intubation feasibility. Head Neck.
- Truong AT, Truong DT (2015) Retromolar flexible fiber-optic orotracheal intubation: a novel alternative to nasal intubation and tracheostomy in severe trismus. Head Neck 37: 423-424.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences